
5 (tartrazine) sensitivity in the general population is low, it is frequently seen in patients who also have aspirin hypersensitivity. Although the overall incidence of FD&C Yellow No. 5 (tartrazine), which may also cause allergic-type reactions (including bronchial asthma) in certain susceptible persons. Each program has its own eligibility and coverage rules. About twenty-three states and one territory offer these programs to Part D enrollees. If a hypersensitivity reaction occurs, discontinue the drug and initiate appropriate therapy. SPAPs are state-funded programs that provide low-income and medically-needy senior citizens and (sometimes) individuals with disabilities financial assistance for prescription drugs. Allergic Reactions: Hypersensitivity reactions, including anaphylaxis, angioedema, bronchospasm, urticaria, and rash have been reported in post-marketing experience, with some resulting in emergency treatment.Careful consideration should be given to this potential risk prior to initiating therapy. Ischemic Heart Disease, Arrhythmias, and Congestive Heart Failure: NORTHERA therapy may exacerbate existing ischemic heart disease, arrhythmias, and congestive heart failure.  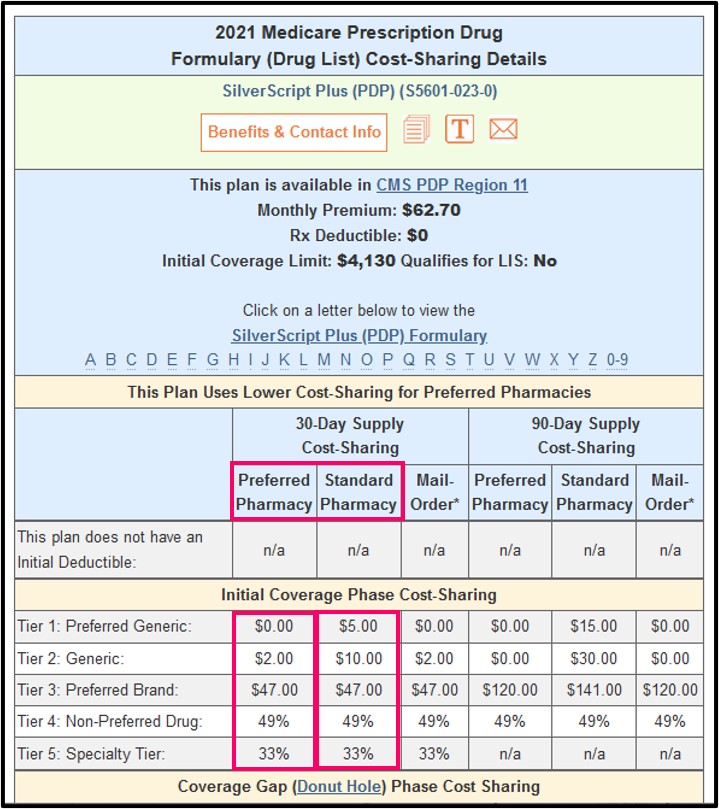
The early diagnosis of this condition is important for the appropriate management of these patients. NMS is an uncommon but life-threatening syndrome characterized by fever or hyperthermia, muscle rigidity, involuntary movements, altered consciousness, and mental status changes. Observe patients carefully when the dosage of NORTHERA is changed or when concomitant levodopa is reduced abruptly or discontinued, especially if the patient is receiving neuroleptics. Hyperpyrexia and Confusion: Cases of a symptom complex resembling neuroleptic malignant syndrome (NMS) have been reported with NORTHERA use during post-marketing surveillance.Supine Hypertension: NORTHERA therapy may cause or exacerbate supine hypertension in patients with nOH, which may increase the risk of cardiovascular events if not well managed, particularly stroke.||The coverage gap (“donut hole”) is a phase of Medicare Part D plans that begins after the total costs spent by both the patient and health plan reach a certain threshold. §HCPs and patients are responsible for complying with applicable plan requirements, and may want to contact a plan directly to confirm its specific requirements. Lundbeck does not control the Medicare program and does not make any guarantees with regard to coverage or Medicare plan terms. ‡Coverage may be subject to PA or trial and failure of prior therapy. State Pharmaceutical Assistance ProgramsĪny information regarding Medicare Part D plans is not intended to imply disease prevalence or appropriate patients for treatment with NORTHERA Lundbeck is not providing this information to influence HCPs’ independent medical judgments regarding patients for whom NORTHERA may be appropriate.Plans that provide supplemental coverage during the coverage gap || If you are eligible for Medicare, you must show proof of enrollment in a Medicare Prescription Drug Plan (Part D) - and in Social Securitys Extra Help Program.Below are 2 resources that may allow eligible Part D patients to lower their costs 4
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
